Hacking the Automatic Detection Process: Finding Eye Cysts with A.I.
Introduction
Cystoid Macular Edema (CME) is a buildup of fluids in the eye near the macula of the eye that can distort a person’s vision. Optical coherence tomography (OCT) is one of the main methods by which physicians detect these edemas.
Automatic detection and quantification of the cysts allows researchers to accurately ascertain the effectiveness of different treatments, which results in more precise and efficient research and treatment.
We have developed a way of detecting and quantifying CMEs that is faster and more accurate than current industry-accepted methods. Read on and let us know what you think.
Accepted Framework:
Current attempts to create an automatic detection system usually follow this main framework:
- Automatic denoising based on prior knowledge (Either of the OCT process and characteristics or of the specific OCT machine in use) or generalized denoising (such as automatic kernel regression)
- Pre-processing – In some of the papers there is an attempt to align the 3D scan, usually based on finding the RPE layer of the retina and setting it as the anchor point around which the images should be aligned. This process is called “Retina flattening”
- Layer segmentation – The retina can be viewed as a stack of different physical layers, each with different light-reflecting properties which allows us to differentiate between them in the scans. Most systems are centered around the correct segmentation of the layers into as many as 11 different layers
- Cyst detection – using the previously segmented layers the region of interest (ROI) for searching the cysts is defined and, at least in the more current research, it is used to learn textural and other features to be used in the detection of the cysts themselves.
RSIP Vision’s Approach
Our approach is somewhat different (see also the linked pages for more details):
- Denoising: Recognizing that efficient denoising that will simultaneously enhance layers as well as respect the cysts’ boundaries is a very difficult mission, we developed two separate denoising algorithms – one aimed at enhancing and smoothing the layer borders for the layer segmentation and the other aimed at reducing speckle noise while retaining as much of the structural integrity of the cysts.
- Selective image re-alignment: taking the same approach by which there is no ‘one method catches all’ for each of the following steps we aligned the images according to the step’s distinct need. While for segmenting the layers we did flatten the retina based on the RPE layer, for detecting the seed points for the cysts we aligned the images based on the ILM and for the final cyst detection one of the algorithms we propose uses no flattening at all.
- Layer segmentation: Finding the RPE and ILM layers using Bellman-Ford’s algorithm for path finding using the intensity gradient as a cost function. After aligning the retina based on the RPE we just found, we used the Bellman-Ford algorithm iteratively starting from the top of the retina. We locate the layers using the intensity gradient as cost function and reducing the search ROI.
- Seed detection: instead of jumping straight to cyst detection we first try to find key points that we can with greater certainty determine belong to cysts and then use them to segment the entire cyst. We use a machine-learning based algorithm which uses super-pixels to further segment the ROI and return the relevant super-pixels as the cyst seed. The algorithm is based on an ensemble of random forests with AdaBoost for feature selection.
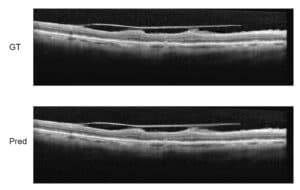
- Cyst detection: Finally, using the incriminating seed super-pixels found we propose two algorithms to detect the actual cysts. The first algorithm uses a graph-cut algorithm and the relations between a cyst and its surroundings to find the actual boundaries of each cyst. The second algorithm uses convolutional neural networks for deep learning of the cysts’ textural features and automatic recognition, ensuring continuous detection training and improvement.
Together the steps proposed by RSIP Vision allow for more efficient automatic detection and analysis of Cystoid Macular Edema than other usual methods. This in turn allows for faster research developments, and ultimately, better treatment for CME.
NB: image above is by Rebellos (own work) [CC BY-SA 3.0]

 Ophthalmology
Ophthalmology